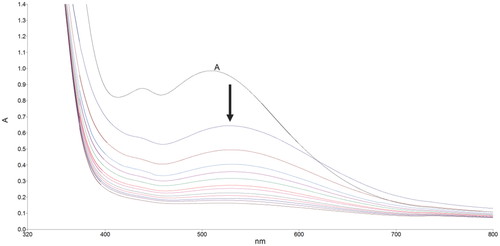
Kinetics of acid‐catalyzed hydrolysis of some high‐spin Fe(II) Schiff base amino acid complexes were followed spectrophotometrically at 298 K under pseudo–first‐order conditions. The studied ligands were derived from the condensation of 5‐bromosalicylaldehyde with different four amino acids (phenylalanine, aspartic acid, histidine, and arginine). The acid hydrolysis reaction was studied in aqueous media and in the presence of different concentrations of the alkali halide (KBr) and cationic surfactant (cetyl‐trimethyl ammonium bromide, CTAB). The general rate equation was suggested to be rate = kobs[complex], where kobs = k2[H+]. The increase in [KBr] enhances the reactivity of the reaction, and the addition of CTAB to the reaction mixture accelerates the reaction reactivity. The obtained kinetic data were used to determine the values of δmΔG# (the change in the activation barrier) for the studied complexes when transferred from “water to water containing different [KBr]” and from “water to water containing altered [CTAB].”