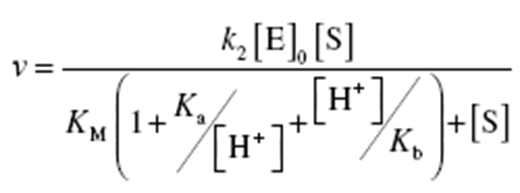Chemical kinetics 321 C for third year students - chemistry Program
Content
1.Rates of complex reactions (Rev.)
2.Simplification of kinetic schemes
3.Reactions in Solutions
4.Reactions on Surfaces
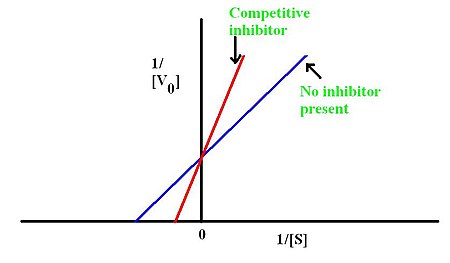
5.Enzymatic Catalysis
6.Acid–Base Catalysis
7.Autocatalysis