Kinetic Theory
Course Content
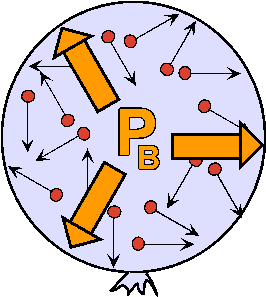
-The kinetic Model of Gases.
-The Pressure of a Gas.
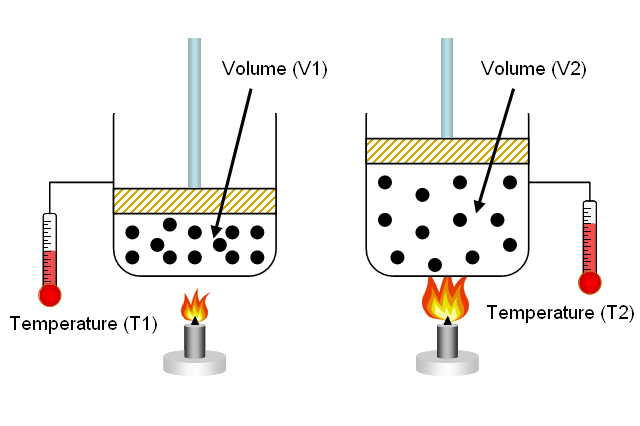
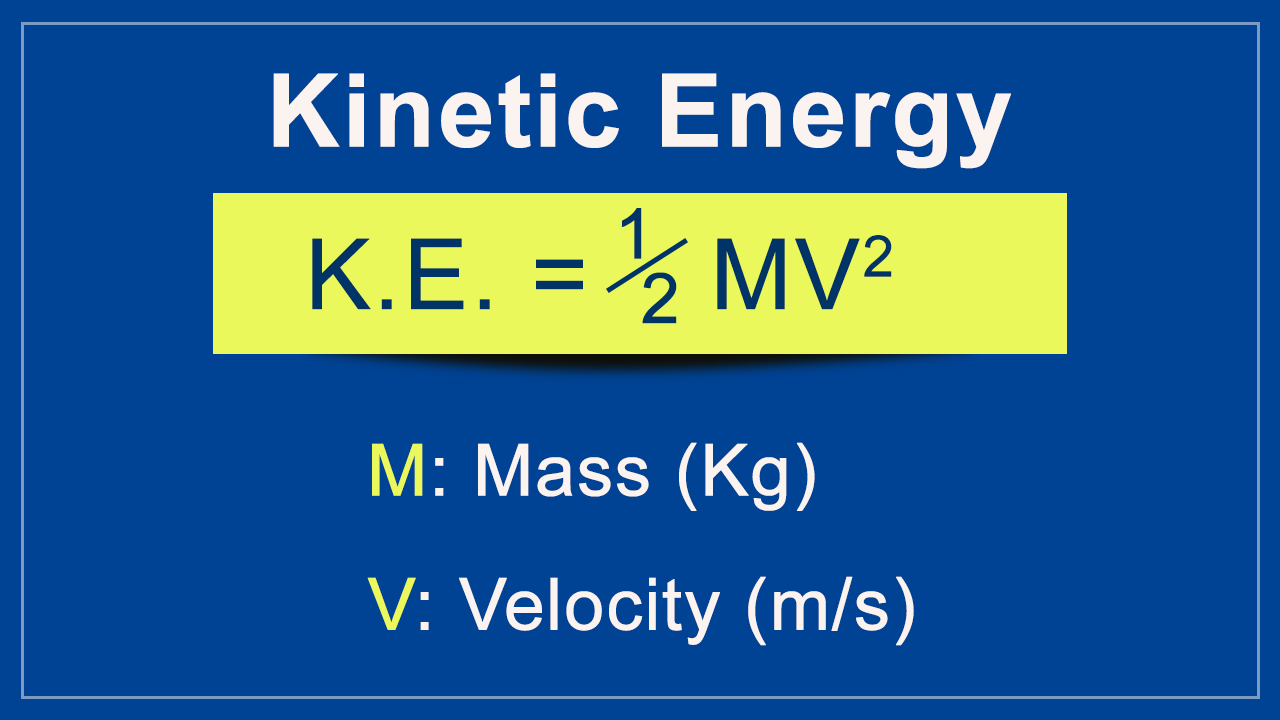
-Molecular Velocities, Kinetic Energies and Temperature.
-Numerical Values for Molecular Energies And Molecular Speeds.
-Degrees of Freedom.
- Applications of the Gas Laws.
-The Distribution of Molecular Speeds along One Direction.
-The Distribution of Molecular Speeds in Three-Dimensional space.
-The Mean Free Path, Collision Diameter, and Collision Numbers .
-The Kinetic Theory of Gas Viscosity.
-Theory of Non-ideal Behavior.
-Van der Waals' Equation.
- Effusion and Diffusion of Gases.
-Van der Waals' Equation and the Critical Point.