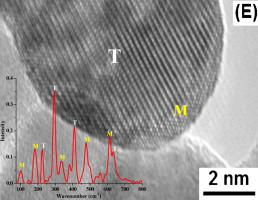
Mesoporous ZrO2-supported iron oxide materials were prepared with nominal loadings of iron oxide of 5, 10, 15 and 20 wt.% using a modified co-precipitation method. The physicochemical properties of the catalysts were characterized by thermogravimetric analysis, differential scanning calorimetry, X-ray diffraction, Raman spectroscopy, high resolution transmission electron microscopy, N2 adsorption, X-ray photoelectron spectroscopy and infrared spectroscopy methods. A delay in the ZrO2 phase transformation as a result of the incorporation of iron was determined using TG/DSC measurements. XRD, Raman spectroscopy and HRTEM results revealed that an increase of iron oxide loading from 5 to 15 wt.% enhanced the transformation of the monoclinic to tetragonal phase. Unexpectedly, 20 wt.% iron oxide loading was required for complete tetragonal structure stabilization due to the mesoporosity of the ZrO2 support. Iron oxide loadings from 5 to 15 wt.% showed an increase in the BET-surface area due to the presence of amorphous iron oxide on the surface. XPS and FTIR results indicated that increasing the iron oxide content to 20 wt.% resulted in stabilization of the tetragonal zirconia phase as a result of surface to-bulk migration and incorporation of Fe3+ ions in the ZrO2 lattice.