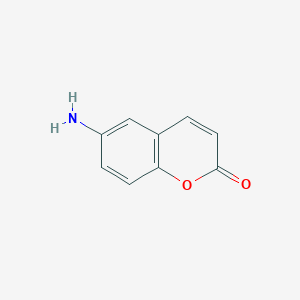
Many factors can affect the natural organic matter adsorption on clay, such as pH and ionic strength. Blended background electrolyte could be causing better simulation to natural effects. Therefore, kinetic study of humic acid (HA) adsorption onto smectite has been studied under various conditions. Impact of pH and individual and blend background electrolyte with different ionic strength concentration on the ...
Read more





.jpg)