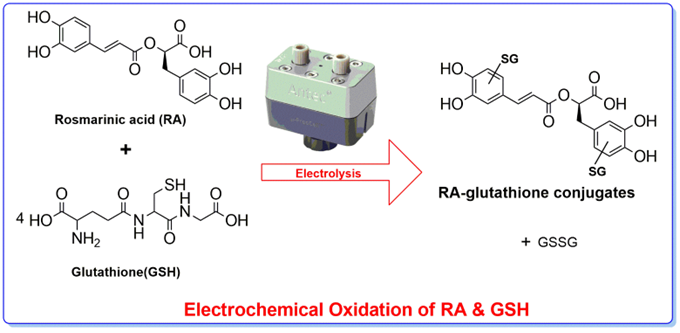
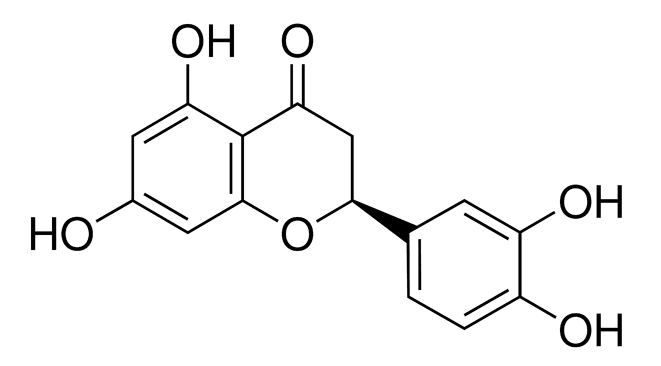
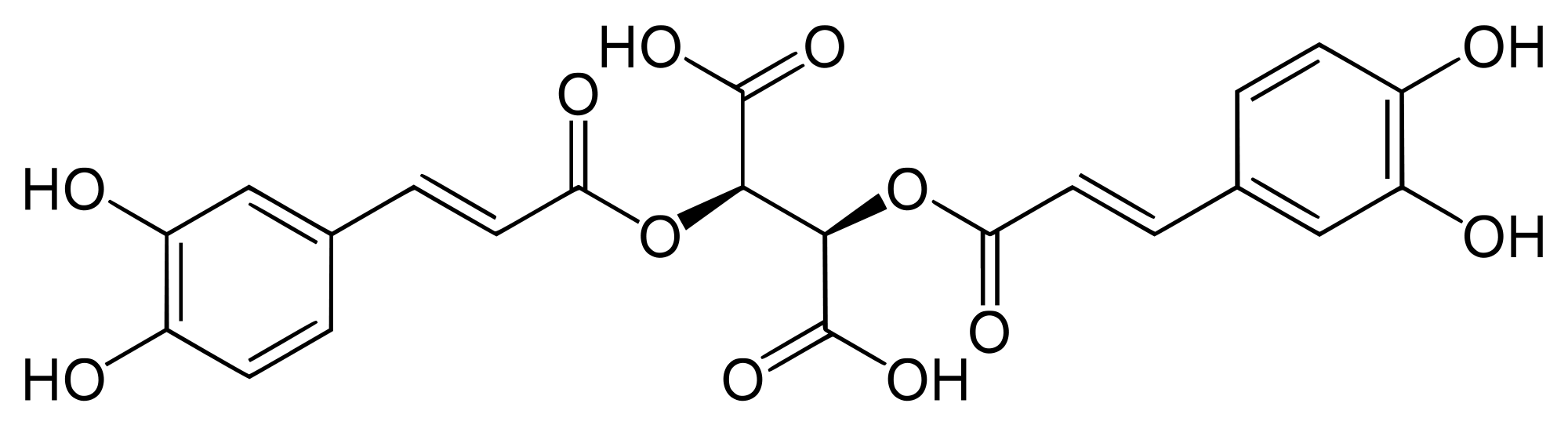
Natural polyphenols are omnipresent and are an integral part of the human diet as well as quinones. Glutathione (GSH) is present in a significant amount inside cells and consequently, GSH conjugates of polyphenols will be encountered in the body. In the current work, voltammetry and liquid chromatography–mass spectrometry were carried out to characterize the reaction mechanism of the electrochemical oxidation ...
إقراء المزيد