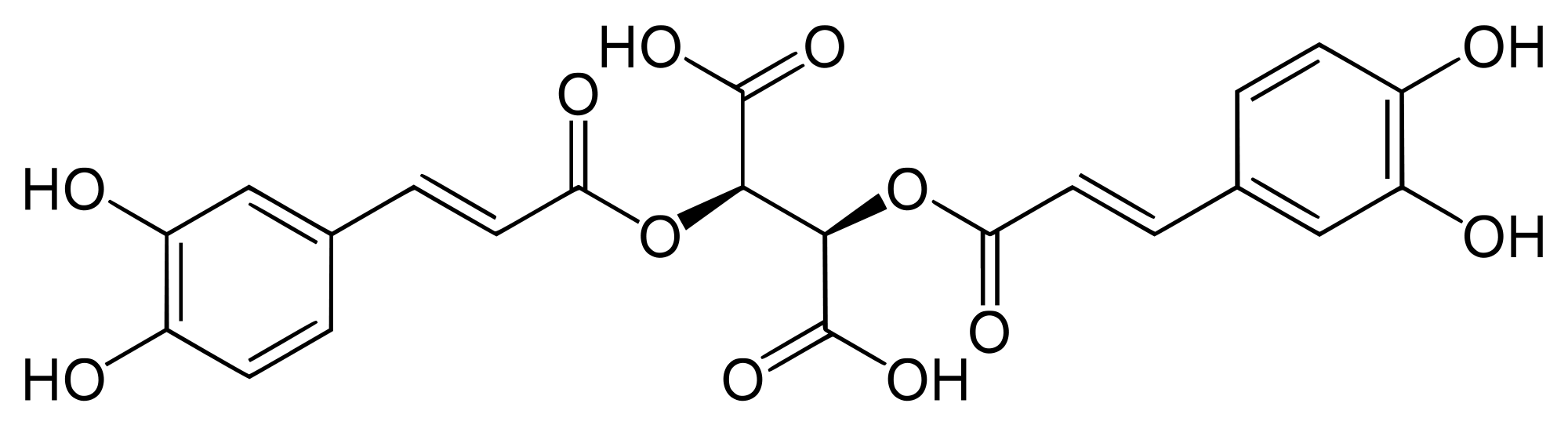
Electrochemical oxidation of chicoric acid (ChA) was investigated using cyclic voltammetry and chronoamperometry at a glassy carbon electrode. Chicoric acid generates single quasi‐reversible redox wave in cyclic voltammetry over a wide pH range, and an ECEC‐dimerization mechanism is proposed. Effect of glutathione (GSH) on the electrochemical oxidation of chicoric acid (ChA) was investigated in Britton−Robinson buffer solution. Ultra‐high performance liquid chromatography (UPLC) coupled with mass spectrometry (MS) was used to show that the naturally occurring chicoric acid (ChA) underwent an electrochemical oxidation in the presence of glutathione (GSH) to form mono‐, bi‐, tri‐, and four‐glutathione conjugates of chicoric acid and a mono‐glutathione conjugate of a chicoric acid dimer. The obtained results are useful for understanding and predicting the oxidative degradation pathway of chicoric acid.