The cationic model of NO is isoelectronic with CO in formation of ……………
[a- σ bond b- π donation c- both a and b d- none] with the metal.
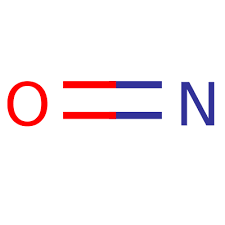
The cationic model of NO is isoelectronic with CO in formation of ……………
[a- σ bond b- π donation c- both a and b d- none] with the metal.